Tag: American Medical Association (AMA)
Goverment Shutdown: What you Need to Know
With the government shutdown in effect, the Texas Medical Association is in regular communication with federal colleagues at the American Medical Association on the impact to physician practices, as AMA remains in touch with senior officials at the Centers for Medicare & Medicaid Services (CMS).
The volatility in Washington, D.C. could mean the shutdown lasts days to weeks, and TMA will share additional information via Texas Medicine Today as the situation develops. Here is what physicians need to know so far:
Medicare/Medicaid: The CMS contingency plan states that during a lapse in funding, the Medicare program will continue. CMS also says it has sufficient funding for Medicaid to fund the first quarter of 2026 and is maintaining the staff necessary to make payments to eligible states for the Children’s Health Insurance Program.
Medicare Administrative Contractors (MACs) have been instructed to implement a temporary claims hold of 10 business days, which AMA says should have minimal impact on physicians due to the 14-day payment floor. “The hold prevents the need for reprocessing large volumes of claims should Congress act after the statutory expiration date,” states a CMS Medicare Learning Network Connects special edition email newsletter that, as of this writing, had not yet been publicly posted. Physicians may continue to submit claims during this period, but payment will not be released until the hold is lifted. For the latest information, AMA recommends physicians monitor their MAC’s website (Novitas in Texas) and this CMS webpage.
“In the past, Congress generally has restored lapsed policies back to the effective date of the shutdown. During the shutdown, the AMA is monitoring any potential delays in Medicare claims processing or other Medicare payment problems that could result from federal staffing reductions at CMS, including during the shutdown,” said AMA CEO and Executive Vice President John Whyte, MD, MPH.
Telehealth: Physicians who provide telehealth services to fee-for-service Medicare patients should be aware that the Medicare telehealth flexibilities lapsed on Sept. 30 for most care and congressional action is required to restore that waiver. The exceptions are patients being treated for mental health and/or behavioral health disorder (including substance use disorders), stroke, and monthly end-stage renal disease visits for home dialysis. Otherwise, telehealth services are limited to rural areas as they were before the COVID public health emergency, and patients cannot receive telehealth services in their homes. The ability to provide audio-only services to Medicare patients also lapsed, as did the Acute Hospital Care at Home program.
The CMS newsletter suggests “practitioners who choose to perform telehealth services that are not payable by Medicare on or after Oct. 1, 2025, may want to evaluate providing beneficiaries with an Advance Beneficiary Notice of Noncoverage.”
However, AMA notes physicians in certain Medicare Shared Savings Program accountable care organizations can continue to provide and be paid for telehealth services.
Other services: CMS says it is continuing federal insurance marketplace activities, such as eligibility verification. Other nondiscretionary activities including health care fraud and abuse control and Center for Medicare & Medicaid Innovation activities are also continuing.
However, AMA reports that funding extensions for community health centers, the National Health Service Corps, and teaching health centers that operate graduate medical education programs expired on Sept. 30, and the 1.0 work geographic practice cost index floor extension expired on Oct. 1. Additional programs that have lapsed include: special diabetes programs; public health emergency authorities (e.g., Public Health Emergency Fund); increased inpatient hospital payment adjustment for certain low-volume hospitals; the Medicare-Dependent Hospital program; quality measure endorsement, input, and selection; and outreach and assistance for low-income programs (e.g., area agencies on aging).
TMA wants to hear from you. If you or your practice staff experience Medicare payment delays, telehealth obstacles, or other concerns, please let us know through TMA’s Knowledge Center to help inform advocacy.
Amy Lynn Sorrel
Associate Vice President, Editorial Strategy & Programming
Division of Communications and Marketing
Is Your Plate Full?
Saving Time Playbook
This playbook will provide strategies to organizational leaders to enhance processes, implement time-saving workflows, increase the efficiency of their practice, and improve physician well-being
POINTS OF INTEREST:
- The Burnout Problem Is Organizational, Not Personal
- Stop the Unnecessary Work
- Getting Rid of Stupid Stuff
- Share the Necessary Work
- Advanced Rooming and Discharge
- Tasks Medical Assistants or Other Team Member Can Complete
- Make the Case to Leadership
- And Much, Much More: Graphs, podcast, practical tools, checklist, and toolkits
COVID-19 CPT vaccine and immunization codes

After the 2024 CPT book was printed, the AMA deleted 64 codes and Appendix Q.
COVID-19 Vaccines
Administration
• 90480 – Immunization administration by intramuscular injection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) vaccine, single dose
Vaccine Product
• 91318 – Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) vaccine, mRNA-LNP, spike protein, 3mcg/0.3 mL dosage, tris-sucrose formulation, for intramuscular use (6
months to 4 years) Pfizer
• 91319 – Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)(coronavirus disease [COVID-19]) vaccine, mRNA-LNP, spike protein, 10mcg/0.3 mL dosage, tris-sucrose formulation, for intramuscular use (5-11
years) Pfizer
• 91320 – Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) vaccine, mRNA-LNP, spike protein, 30 mcg/0.3 mL dosage, tris-sucrose formulation, forintramuscular use (12 years and older) Pfizer
• 91321 – Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) vaccine, mRNA-LNP, 25 mcg/0.25mL dosage, for intramuscular use (6 months – 11 years) Moderna
• 91322 – Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) vaccine, mRNA-LNP, 50 mcg/0.5 mL dosage, for intramuscular use (12 years and older) Moderna
More on DEA Training
Please click on this link “DEA Registered-Practioners” to read the Letter from the Drug Enforcement Administration (DEA).
Pay close attention to “Group 2”. There are other ways to satisfy the training requirement.
Additional Links:
How’d You Score?
Did you take Betsy Nicoletti’s Quiz on Modifier 25?
Let’s Explore the Facts or Rules Behind the Answers.
#1. The definition and rules for modifier 25 did not change in 2023. “In the March edition of the CPT Assistant it says that while the rules weren’t changed in the 2023 code set “confusion exists regarding its appropriate use.”
#2. What does a modifier added to a CPT or HCPCS code do? The answer is b) Indicates the service or procedure was altered by specific circumstances.
Some modifiers affect payment (like modifier 25) and some are informational only (those appended to teaching physician services). CPT modifiers may be used on HCPCS codes and HCPCS modifiers on CPT codes. A modifier doesn’t change the definition of the code, but indicates that the service or procedure was altered in some way.
#3. When using modifier 25 when reporting an E/M services and a minor procedure, do you need a different diagnosis for the E/M and the Procedure? The answer is NO.
“As such, different diagnoses are not required for reporting of the E/M services on the same date”. CMS says, “If a significant separately identifiable evaluation and management service is performed, the appropriate E&M code should be reported utilizing modifier 25 in addition to the chemotherapy administration or nonchemotherapy injection and infusion service. For an evaluation and Management service provided on the same day, a different diagnosis is not required.” Medicare Claims Processing Manual, CH. 12 30.5.C
#4. When performing a minor procedure, usual preoperative and postoperative activities are included i in the work of the procedure and should not be reported with an E/M service. Which of these activities are considered usual preoperative and postoperative activities? (List may not be complete). Answer is d) All of the above.
The March 2023 CPT Assistant lists the services that are considered part of the procedure—paid for within the fee for the procedure—and should not be counted as a separate E/M.
Pre- and post-operative services typically associated with a procedure include the following and cannot be reported with a separate E/M services code:
• Review of patient’s relevant past medical history,
• Assessment of the problem area to be treated by surgical or other service,
• Formulation and explanation of the clinical diagnosis,
• Review and explanation of the procedure to the patient, family, or caregiver,
• Discussion of alternative treatments or diagnostic options,
• Obtaining informed consent,
• Providing postoperative care instructions,
• Discussion of any further treatment and follow up after the procedure
5) When performing a preventive medicine service or wellness visit, the practitioner also assesses and manages the patient’s chronic conditions, reviews labs, and renews multiple prescriptions. How is this billed? The Answer is b) Preventive medicine or wellness visit and a problem-oriented visit, with modifier 25 on the problem-oriented visit
Medicare Claims Processing Manual, Ch 12. 30.6.1.1.2.H
“When the physician or qualified NPP, or for AWV the health professional, provides a significant, separately identifiable medically necessary E/M service in addition to the IPPE or an AWV, CPT codes 99201 – 99215 may be reported depending on the clinical appropriateness of the circumstances. CPT Modifier -25 shall be appended to the medically necessary E/M service identifying this service as a significant, separately identifiable service from the IPPE or AWV code reported (HCPCS code G0344 or G0402, whichever applies based on the date the IPPE is performed, or HCPCS code G0438 or G0439 whichever AWV code applies).”CPT Professional Ed., 2023, page 32

Cheers to knowledge
Check Your Modifier 25 Knowledge

Our Friend Betsy Nicoletti founder of CodingIntel, says “It’s hard for me to think of a topic that invites so much disagreement between coders and auditors, between practices and payers and with our practitioners.”
She has put together a short little quiz on the topic (below). Tell us how you did.
AMA unveils anticipated E/M changes for facility-based services

The AMA set off its own fireworks during the Independence Day weekend with the early release of new E/M guidelines that will be included in the 2023 CPT manual and take effect Jan. 1, 2023
Aetna 3rd-Party Claim and Code Review
Beginning June 1, 2022, you may see new claim edits.
According to March 1, 2022 Aetna OfficeLink Updates: These are part of our Third-Party Claim and Code Review Program. These edits support our continuing effort to process claims accurately for our commercial, Medicare and Student Health members. You can view these edits on our Availity provider portal.*
You can view any of these edits on Aetna’s Availity® provider portal.
For coding changes, go to:
- Aetna Payer Space
- Resources
- Expanded Claim Edits
Except for Student Health, you’ll also have access to Aetna’s code edit lookup tools. To find out if our new claim edits will apply to your claim, log in to our Availity provider portal. You’ll need to know your Aetna® provider ID number (PIN) to access our code edit lookup tools
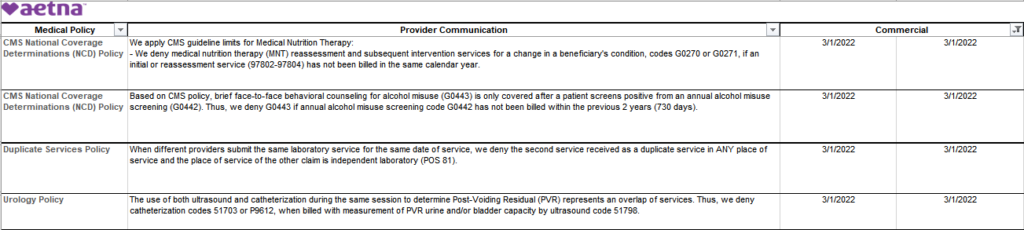
Here is what POET found with 2022 effective dates:
AMA Toolkit Dissects Federal Surprise Billing Law

Much of the federal government’s solution to resolve certain out-of-network billing disputes without balance billing or otherwise involving patients – known as the No Surprises Act – took effect at the start of 2022.
Among other pieces physicians must familiarize themselves with, the new federal law features an independent dispute resolution (IDR) process that was intended to let physicians and insurers both make their case for fair payment. Naturally, plenty of minutiae and arcana exists within the law, and a portion of the rules for the IDR process is under a legal challenge from the Texas Medical Association and others in organized medicine.
To help physician practices understand and navigate the new law, the American Medical Association has created a toolkit, Preparing for Implementation of the No Surprises Act. The 20-page toolkit includes information on:
- Operational challenges physicians “will need to address immediately” to be compliant with the law’s new requirements, such as when uninsured and self-pay patients must receive a good-faith estimate of charges before they receive services;
- What services and care fall under the rules of the No Surprises Act;
- Timetables and requirements for the IDR process; and
- When and how facilities and physician practices can obtain a patient’s consent to balance bill for out-of-network care at an in-network facility.
AMA says it will update the toolkit “as additional guidance is available” and will develop new resources on parts of the law not already included in the toolkit.
For additional information on the No Surprises Act, you can check out TMA’s list of resources on the law, which has both similarities and differences to Texas’ IDR law governing state-regulated health plans.
Meanwhile, TMA and others are still pushing to ensure the implementation of the law is fair for physicians seeking to get paid. In late October 2021, TMA filed suit to challenge what physicians and hospitals say is an unfair piece of the IDR process outlined in federal rules. Check future editions of Texas Medicine Today for updates on that lawsuit.

Recent Comments